Finasteride is an FDA-approved treatment commonly used to slow hair loss and promote hair rethickening in men with androgenetic alopecia, commonly known as androgenetic alopecia (male pattern baldness).
In 1974, a researcher discovered that some Caribbean children had a genetic mutation affecting the enzyme 5-alpha reductase, which prevents the conversion of testosterone into dihydrotestosterone (DHT). As these children matured, they had smaller prostate sizes and lacked male pattern baldness. Building on this discovery, researchers developed finasteride to block the 5-alpha reductase enzyme, reducing DHT levels. In 1992, the FDA approved a 5 mg dose of finasteride (Proscar®) for treating prostate enlargement. Since DHT is involved in both prostate enlargement and hair loss, clinicians observed hair regrowth in patients using finasteride for prostate issues. In 1997, the FDA approved a 1 mg dose of finasteride (Propecia®) for treating male pattern baldness. After the patent expired in 2013, generic versions became widely available, making the treatment more accessible and affordable.
Finasteride is widely prescribed in the United States and remains one of the most commonly prescribed medications for hair loss and prostate enlargement. It ranks 73rd in total prescriptions, with over 9 million prescriptions filled for more than 2.6 million patients. Finasteride usage among men older than 25 years have increased by 200% over the past 7 years.
This growth is driven primarily by the rising prevalence of male pattern baldness, which affects approximately two-thirds of men by age 35. Additionally, increasing awareness through online platforms has contributed to more widespread use of finasteride, making it a popular option for managing androgenetic alopecia.
Off-Label Use
Off-label prescribing is common when new evidence or clinical experience suggests potential benefits not yet included in a drug’s official approval. When a doctor prescribes an off-label medication, they may use it to treat a condition that wasn’t originally approved, recommend a different dosage, or administer it in a new form (such as applying an oral medication topically).
In the early 2000s, physicians began prescribing oral finasteride off-label for treating female pattern hair loss (FPHL), with studies showing that doses between 2.5 and 5.0 mg led to positive results. More recently, since 2017, topical finasteride has also been used to treat both male and female pattern hair loss, demonstrating effectiveness similar to the oral form while potentially reducing the risk of side effects commonly associated with the oral medication.
How Does Finasteride Work?
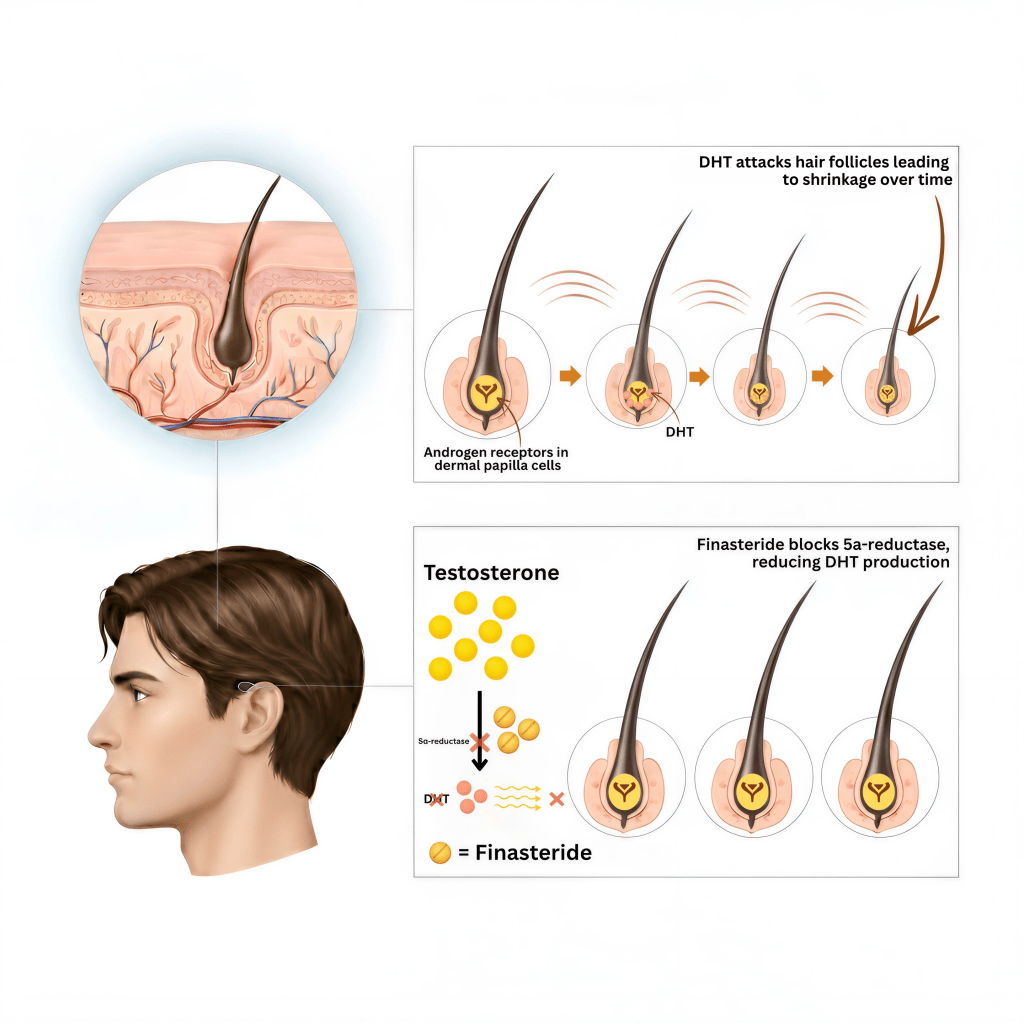
Finasteride acts as a 5-alpha reductase inhibitor (5-ARI), blocking the enzyme that converts testosterone into dihydrotestosterone (DHT). It specifically targets the type II 5-alpha reductase enzyme, which is found in higher concentrations around the hair follicles of men with androgenetic alopecia. By reducing the amount of DHT affecting the hair follicles, finasteride slows the progression of hair loss and makes rethickening possible.
Research shows that taking 1 mg of oral finasteride daily can lower DHT (a hormone linked to hair loss) levels in the blood by 72% and in the scalp by 64%. However, taking a higher dose than 1 mg won’t make a big difference in further reducing DHT. Since finasteride doesn’t block all of the DHT, many people find that while it slows hair loss, it doesn’t completely stop it. Additionally, because the liver metabolizes finasteride, extra caution is needed when prescribing it to patients with liver issues. As with most hair loss treatments, finasteride results will gradually fade, and hair loss typically resumes within 12 months after stopping the medication.
When taken orally, approximately 65% of finasteride is absorbed into the bloodstream, and food does not impact how well it is absorbed. Most of the absorption occurs within 1 to 2 hours after taking the medication, with the process being nearly complete 6 to 8 hours after the dose. DHT levels return to their original levels about 14 days after stopping the medication. This means the effects of the treatment are temporary, and consistent use is often necessary to maintain results.
What is the Difference Between Testosterone and DHT?
Testosterone is the primary male sex hormone (androgen), essential for overall health and well-being in both men and women. It supports energy levels, mood, libido, cognitive function, and physical attributes such as bone strength, muscle growth, and fat distribution. In men, testosterone levels are about ten times higher than dihydrotestosterone (DHT) levels. Excessive testosterone can lead to aggressive behavior, acne, and testicular shrinkage, while significantly lowering testosterone can reduce energy, impair sexual function, and decrease muscle mass.
DHT is a byproduct of testosterone, formed when the 5-alpha reductase enzyme converts testosterone into DHT. It is up to ten times more potent than testosterone and binds more strongly to androgen receptors in hair follicles. DHT is responsible for male characteristics during puberty, such as a deeper voice, facial and body hair growth, and prostate gland development. It also affects skin health by regulating sebum (skin oil) production. High DHT levels play a major role in androgenetic alopecia (male and female pattern hair loss) by shrinking hair follicles, leading to thinning hair and eventual hair loss. Therefore, lowering DHT levels can improve prostate health, lessen oily skin, and reduce hair loss. Although women produce smaller amounts of testosterone and DHT, these hormones still influence their hair, skin, sexual function, bone strength, and energy levels.
Finasteride’s Effect on Testosterone and Estrogen
When finasteride blocks the type II 5-alpha reductase enzyme, it reduces the conversion of free testosterone into dihydrotestosterone (DHT), leading to lower DHT levels. As a result, more testosterone remains in the bloodstream, which may cause a temporary 9-15% increase in testosterone levels, although this increase typically remains within the normal range overall.
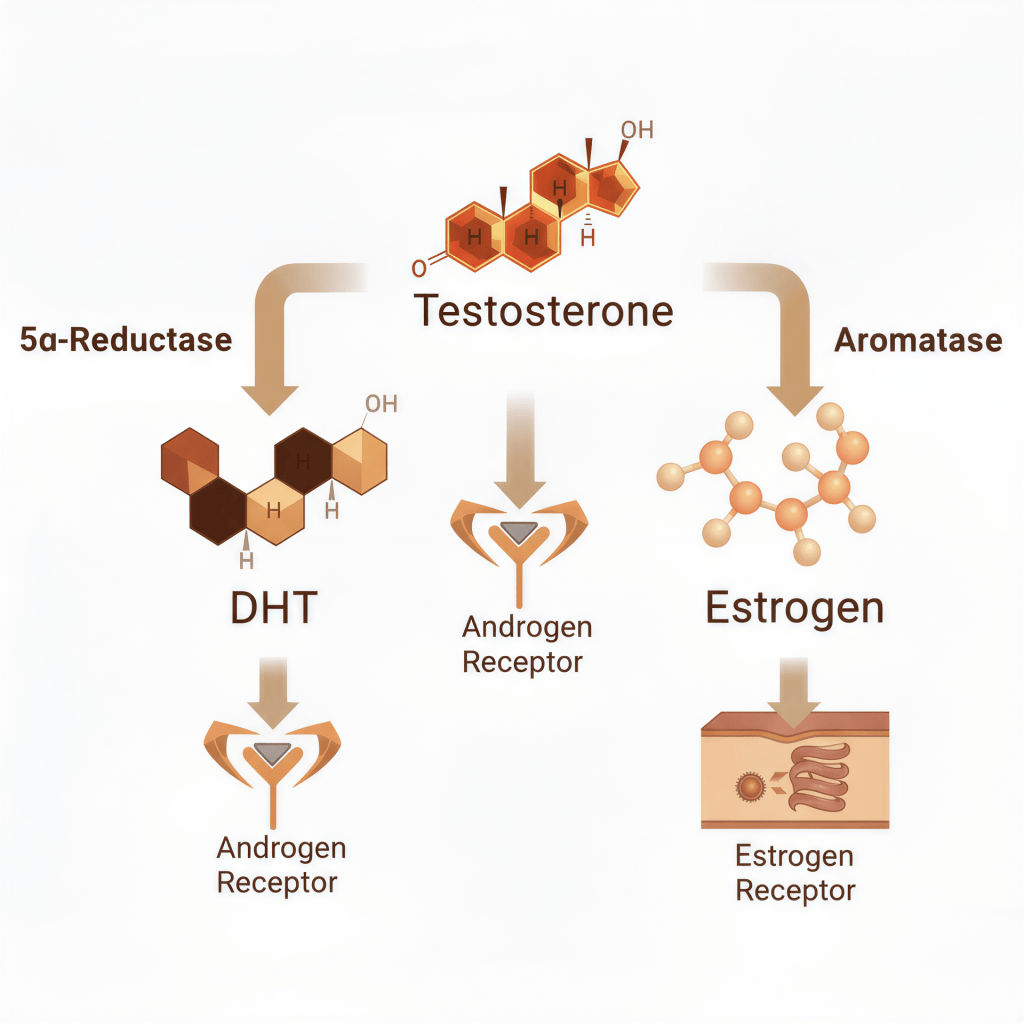
This temporary rise occurs because the body produces the same amount of testosterone, but less is converted into DHT. Over time, the body’s natural negative feedback system adjusts by slightly reducing testosterone production to maintain hormonal balance. Finasteride does not significantly alter total or free testosterone levels in the long term, ensuring a stable hormonal environment.
Because finasteride’s DHT-blocking action temporarily increases free testosterone levels, a small amount may be converted to estradiol, a form of estrogen, through a process called aromatization.
Although the increase in estrogen levels is typically minimal, some men might notice mild estrogenic side effects such as breast tenderness or slight breast enlargement (gynecomastia). However, these side effects are uncommon and occur in less than one percent of users. For most people, the rise in estrogen is not clinically significant and doesn’t lead to major hormonal imbalances.
Topical Finasteride
While topical finasteride has only recently gained popularity, the first significant study by Mazzarella et al. in 1997 showed promising results for hair regrowth and reducing hair loss without reported side effects. Over time, more research has validated its effectiveness in treating androgenetic alopecia (pattern hair loss), confirming its safety and efficacy.
Merck, the company that makes Propecia® (brand name oral finasteride), decided not to seek FDA approval for a topical version of the drug. This choice was likely due to several reasons, including the success of the oral version, the high cost of developing new drugs, and uncertainty about how well a topical version would work. As a result, topical finasteride has primarily been driven by other companies and compounded by pharmacies for off-label use. Since each pharmacy may make its own version, it can be hard to know how effective a particular formulation will be.
Topical finasteride is typically applied once daily to the scalp, with concentrations ranging from 0.005% to 1%, and is often combined with topical minoxidil to enhance effectiveness. This option appeals to those concerned about potential side effects from oral finasteride, as topical application minimizes systemic absorption.
Like its oral counterpart, topical finasteride inhibits the type II 5-alpha reductase enzyme, limiting the conversion of testosterone into DHT. However, it mainly reduces DHT levels in the scalp rather than in the bloodstream, helping to slow hair loss and promote the re-thickening of thinning hair while minimizing the risk of systemic side effects.1
Topical finasteride minimizes systemic absorption and primarily targets the scalp to reduce local DHT production. Studies suggest topical finasteride has lower systemic bioavailability than oral, leading to fewer side effects while still maintaining similar efficacy in treating hair loss by reducing scalp DHT levels. However, the exact bioavailability varies based on formulation and application.2,3,4
The advantages of topical finasteride include:
- Reduced Systemic Absorption: Since topical finasteride is applied directly to the scalp, it minimizes the drug’s presence in the bloodstream, possibly lowering the risk of systemic side effects commonly associated with oral finasteride
- Better Tolerability: Men who experienced side effects from oral finasteride have successfully tolerated the topical version
- Alternative for Non-Responders: Some men who did not respond to oral finasteride have shown improvement with the topical formulation, especially when combined with minoxidil
Finasteride’s Effectiveness for Hair Loss
Both oral and topical finasteride are effective options for treating androgenetic alopecia (AGA), a common form of hair loss in both men and women. Choosing between the two often depends on your personal preferences, weighing the potential benefits against any risks, your specific medical needs, and compliance. Large clinical trials have shown that finasteride works well for men with hair loss caused by androgens, particularly in those with mild to moderate thinning at the middle of the scalp and crown.
Finasteride Dosage and Efficacy
Research by Drake et al. found that a daily dose of 0.2 mg of oral finasteride was nearly as effective as 1 mg in reducing DHT levels in both the scalp (56.5%) and the bloodstream (64%). The study further demonstrated that DHT levels in the scalp (64% vs. 69%) and bloodstream (71% vs. 72%) were similar between daily doses of 1 mg and 5 mg, indicating that higher doses do not lead to significant clinical benefits. While a 0.2 mg dose can still provide some benefits, a 1 mg daily dose is necessary to achieve the most effective reduction of DHT levels and optimal hair preservation.
Long-Term Finasteride Benefits
Kaufman et al. and Layden et al. showed that taking 1 mg of finasteride daily significantly slows hair loss in men with androgenetic alopecia and promotes hair regrowth in many cases. After one year of use, 48% of men experience regrowth, increasing to 66% after two years. Notably, 83% of men who took finasteride for two years saw no further hair loss.
The previous two studies were extended to 5 years, with 83% of men with crown (vertex) hair loss maintaining their hair after two years, and 70% of men with frontal hair loss had no further loss after one year. Visible crown regrowth occurred in 61% of men after two years, while 37% showed frontal area regrowth after one year. After five years, 65% of men treated with finasteride had more hair than they did at baseline. An expert panel found that 90% of finasteride-treated men either maintained their hair or experienced visible improvement over five years, compared to 75% of placebo-treated men who experienced worsening hair loss. No new side effects were reported during these trials, and any side effects reported in the first year decreased over time with continued use.
Rossi et al. conducted a 10-year study on finasteride’s long-term efficacy. Most participants saw initial improvements after the first year, and their condition continued to improve. The positive trend was maintained, and finasteride’s benefits peaked after 2-3 years but lasted for at least 5-10 years. Even if some hair loss resumed after this period, it progressed much more slowly than those not using finasteride. Notably, 75% of those who did not receive finasteride experienced significant hair loss within five years. Men who used the medication for ten years retained significantly more hair than those who did not.
In a 2006 study, 708 men with androgenetic alopecia (AGA) took 1 mg of finasteride daily for about four months. The results showed that 91.8% of the men experienced improvement in their hair growth.
In this study, 87.1% of patients (2,230 out of 2,561 patients) experienced some level of hair growth improvement, ranging from slight to significant increases.
Oral Finasteride’s Efficacy in Women
Oral finasteride is used off-label for female pattern hair loss (FPHL). Studies have shown that women responded best to 2.5 and 5 mg dosages with reduced hair loss and improved hair density. Because FPHL is a complex disease, finasteride is generally less effective in women than men. However, it is still proven to be a worthwhile treatment option.
A notable 3-year study found that when 1.25 mg of finasteride was administered daily to women with androgenetic alopecia, 82% of participants experienced a significant increase in hair thickness.
A meta-analysis revealed that 81% of women taking less than 2.5 mg oral finasteride daily and 86% taking 2.5 mg or more had an improved hair response. This suggests that while higher doses of finasteride may offer marginally better results, even lower doses are highly effective for most women with androgenetic alopecia.
A 24-week study examining the effects of different treatments for female-pattern hair loss found that taking 5 mg of oral finasteride daily resulted in the most significant improvement in hair density. This dosage outperformed other treatments, such as 5% topical minoxidil (applied once or twice daily), 1 mg oral minoxidil, and lower doses of both minoxidil and finasteride. The results highlight finasteride’s effectiveness in treating hair thinning in women when used at higher doses than other common treatments.
Topical Finasteride Studies
Topical finasteride is formulated to deliver the benefits of finasteride directly to the scalp, reducing dihydrotestosterone (DHT) levels locally while minimizing systemic absorption and the associated risk of side effects.
The first study on topical finasteride in humans, conducted by Mazarella et al. in 1997, involved men and women with androgenetic alopecia. These patients were randomly assigned to use either a 0.005% topical finasteride solution or a placebo twice daily for 16 months. By six months, those using topical finasteride showed a considerable reduction in hair loss compared to the placebo group, and 73% of finasteride users reported “high effectiveness.”
In recent years, topical administration of finasteride has been studied even further. A systematic review, which included seven studies, was performed to evaluate the treatment of AGA with topical finasteride. Researchers found that in patients treated with topical finasteride for AGA, there was a significant reduction in the rate of hair loss, an increase in total hair counts, and positive hair growth assessments in all studies.
Topical Finasteride’s Efficacy in Women
Topical finasteride is also used off-label for treating FPHL. While evidence is still emerging, initial studies and clinical experiences suggest that topical finasteride can effectively reduce hair thinning and promote hair density in women, particularly in the central scalp area.
Nobari et al. performed a published literature search from 1999 to 2020 and found several articles that met specific criteria. The results suggest that a daily dose of 5 mg oral finasteride can be an effective and safe treatment for women with FPHL, especially when combined with topical minoxidil. Additionally, topical finasteride was more effective than other topical treatments for hair loss.
A study comparing 0.25% topical finasteride combined with 3% minoxidil to 3% minoxidil alone found that the combination significantly improved hair density and diameter.
Comparative Study on Topical and Oral Finasteride in Androgenetic Alopecia
The same systematic review involving seven studies evaluated the efficacy of oral versus topical finasteride in treating androgenetic alopecia (AGA). The findings revealed that topical finasteride exhibited comparable effectiveness to oral finasteride in promoting hair regrowth. This suggests that topical formulations can offer similar benefits to oral treatments while potentially minimizing the risk of systemic side effects, making it a viable alternative for patients concerned about the oral route.
In a 2021 study by Piraccini et al., topical finasteride significantly improved hair counts compared to a placebo, demonstrating results similar to oral finasteride. However, the study highlighted that topical finasteride resulted in lower systemic absorption and decreased bloodstream DHT levels compared to the oral form (34.5% vs. 55.6%). Although systemic side effects linked to reduced bloodstream DHT may still occur with topical finasteride, this study suggests a lower risk than oral finasteride, making it a promising alternative for individuals concerned about systemic side effects.
Finasteride versus Minoxidil Studies
This study comparing 1 mg oral finasteride and 5% topical minoxidil found that oral finasteride was significantly more effective in treating androgenetic alopecia, with an 80% success rate versus a 52% success rate for topical minoxidil. This suggests that while both treatments are helpful, oral finasteride delivers more robust results in halting hair loss and promoting regrowth in male pattern baldness. These results highlight finasteride’s potency in reducing DHT levels, a key factor in male hair loss, compared to minoxidil, which works through a different mechanism, primarily enhancing blood flow to hair follicles.
In a comparative study of 450 men, 59% using 5% topical minoxidil and 80.5% using oral finasteride 1 mg showed hair improvements after a year of continuous therapy.
Combination Finasteride with Minoxidil Studies
Research consistently shows that combining finasteride with minoxidil significantly improves hair counts and maintains healthy hair density, reducing the chances of hair thinning or miniaturization progressing compared to either treatment alone. The dual synergistic action of finasteride reducing DHT (a primary cause of genetic hair loss) and minoxidil stimulating hair growth creates an enhanced effect that optimizes hair results, offering a more comprehensive treatment plan for patients looking to stop hair loss and promote regrowth.
Numerous clinical trials support this synergistic approach and highlight the complementary benefits of using both drugs:
The combination of 0.1% topical finasteride with 3% minoxidil was shown to produce superior results in treating androgenetic alopecia compared to using 3% minoxidil alone, with no reported adverse effects, according to a 2012 study by Tanglertsampan C. In the one-year comparative study of 450 men showing oral finasteride’s effectiveness (80.5%) over 5% topical minoxidil (59%), a third group of participants combining both treatments had the highest improvement rate, with 94.1% showing better hair results. Similarly, a 2015 study by Chandrashekar et al. demonstrated that combining 0.1% topical finasteride and 5% minoxidil helped maintain hair density in 84.44% of patients who had previously used oral finasteride and topical minoxidil for two years.
A 24-week study explored the effects of 0.25% topical finasteride with 3% minoxidil compared to minoxidil alone in men with androgenetic alopecia. The combination therapy led to a 90% improvement rate, with patients reporting moderate to significant hair growth and no systemic side effects. More recently, a 6-month study in 2024 confirmed that combining 5% topical minoxidil with 0.25% topical finasteride was significantly more effective than either treatment alone while maintaining a similar safety profile.
Finasteride for Hair Transplant Surgery
While finasteride is commonly discussed as a medical treatment for those seeking to slow or stop hair loss, it also greatly benefits patients undergoing hair transplant surgery. This study shows that combining hair transplant surgery with 1 mg of oral finasteride significantly improves scalp coverage. This improvement occurs as the non-transplanted hair surrounding the transplanted areas becomes denser, enhancing overall hair density and coverage.
Androgenetic alopecia is a chronic, progressive condition with no cure, meaning hair loss may continue over time. While hair transplant surgery restores hair to bald areas, it does not stop the underlying disease. To minimize further hair loss and maintain the natural appearance of your hair in the long term, using DHT-blocking medications is crucial.
Effects on PSA (Prostate-Specific Antigen) Blood Test
PSA (Prostate-Specific Antigen) levels are used as a screening tool for conditions like prostate enlargement (BPH) and prostate cancer. While elevated PSA levels can indicate prostate cancer, they are often linked to benign conditions such as BPH or prostatitis.
Finasteride, a medication that reduces DHT levels and supports prostate health, can lower PSA levels by approximately 50% after three to six months of use. If you’re taking finasteride, it’s important to inform your doctor before a PSA test so they can adjust your results by doubling the PSA value for accurate interpretation.
Some studies show that topical finasteride may have little to no effect on PSA levels. However, its impact can vary depending on the formulation and how much medication is absorbed into the body. Therefore, it is still recommended to inform your doctor if you’re using topical finasteride to ensure proper evaluation.
Infertility Concerns with Finasteride
The impact of finasteride on fertility has been examined by urologists and dermatologists who prescribe finasteride at various doses for prostate and hair conditions. A big rumor regarding finasteride and family planning is that to conceive, finasteride must be discontinued. Before providing facts on finasteride and fertility, it’s important to review the overall prevalence of infertility in men.
Infertility is usually defined as the inability of a couple to conceive after one year of trying. Studies estimat that approximately 1 in 10 men (or around 10%) experience some form of fertility issue, contributing to difficulties in conception for couples. The male is solely responsible in about 20% of cases and is a contributing factor in another 30% to 40% of all infertility cases.
Male infertility is a common issue, with various contributing factor.
Common causes include:
- Low sperm count (Oligospermia): Insufficient sperm production
- Poor sperm motility: Reduced ability of sperm to move effectively
- Abnormal sperm morphology: Sperm with irregular shapes affecting their ability to fertilize an egg
- Hormonal imbalances: Low testosterone or other endocrine disorders
- Varicocele: Enlarged veins in the scrotum affecting sperm production
- Genetic conditions: Disorders like Klinefelter syndrome
- Medications
- Environmental factors:
- Endocrine-disrupting chemicals (EDCs): Chemicals in plastics (BPA, phthalates), pesticides, and industrial pollutants can interfere with hormone regulation and sperm production
- Air pollution: Exposure to heavy metals, particulate matter, and industrial chemicals is linked to lower sperm quality
- Radiation exposure: Increased use of mobile phones and electronic devices near reproductive organs may contribute to sperm damage
- Lifestyle factors: Smoking, marijuana, alcohol, poor diet, obesity, stress, and exposure to toxins
In other cases, the exact cause of infertility remains unknown.
Medications That Can Lower Sperm Counts
Global Decline in Sperm Counts
Over the past 50 years, sperm counts among men worldwide have declined by approximately 50%. This trend, identified in multiple studies, raises concerns about male reproductive health and potential consequences for global population dynamics.
Studies on the impact of 1 mg oral finasteride on sperm production (volume, count, concentration, and motility) have shown mixed results.5,6 However, in two separate studies by Samplaski et al., involving men who visited infertility clinics, only a small percentage of them were using finasteride. Specifically, just 0.6% in one study and 0.9% in the other were finasteride users, out of populations of 4,400 and 4,287 men, respectively.7,8
Overstreet et al. found that finasteride at a daily dose of 1 mg did not alter spermatogenesis, semen production, or sperm morphology in healthy young men.9
While some studies report no changes in sperm count among finasteride users, others suggest that if a reduction does occur, it typically returns to normal within three months after discontinuing the medication. This evidence indicates that although a temporary decrease in sperm count is possible, it happens at a very low rate and is often related to pre-existing infertility-related conditions rather than finasteride itself.10
Male patients planning to conceive who are using finasteride or dutasteride without any history of infertility or sexual side effects can generally continue treatment safely during family planning. For those with existing fertility issues (azoospermia, oligospermia), switching to a combination of topical finasteride and minoxidil may be a better alternative to oral options. However, individual responses to finasteride can vary no matter the delivery method, with some patients being more sensitive to its effects. Women should not take finasteride during pregnancy.
Sometimes, men with previously unknown low semen counts take oral finasteride while planning to start a family. When they later get a semen analysis, they may mistakenly believe finasteride caused the issue. To avoid this, it’s recommended to get a baseline semen analysis before starting finasteride.
No official consensus statement mandates stopping finasteride for men planning pregnancy. However, experts and guidelines recommend stopping finasteride if:
- The male partner is experiencing subfertility or abnormal semen parameters
The best way to protect your future fertility is to get a semen analysis before starting treatment—this gives you a clear baseline and helps determine if there are already any issues with your sperm health.
Is There a Risk of Birth Defects with Finasteride?
In my practice, many men or their partners express concerns about continuing finasteride while planning a family, worried about the possibility of birth defects. Some men report that their partners forced them to stop taking finasteride until after the baby is born, fearing it could affect the pregnancy. Unfortunately, these men often lose the hair gains achieved from finasteride and continue to lose more hair during the lengthy process of family planning and pregnancy as the medication’s effects wear off.
There is no scientific evidence to suggest that men using finasteride are at a higher risk of fathering a child with genital birth defects. The global rate of birth defects is approximately 1-3%, which is the same for both finasteride users and non-users, meaning the medication does not increase the risk. Daily use of 1 mg oral finasteride has not been shown to have any significant impact on sperm that would lead to birth defects.
Another common concern is that pregnant women handling finasteride tablets might risk birth defects in their unborn male child. Since finasteride reduces DHT, a hormone essential in male genital development, there is a theoretical concern about potential abnormalities. However, no adverse pregnancy outcomes have been reported in women exposed to finasteride. The tablets are film-coated, preventing contact with the active ingredient during normal handling, and skin absorption is negligible. Nonetheless, as a precaution, pregnant women should avoid handling crushed or broken tablets.11
Though no evidence links finasteride to male fetal genital abnormalities, women taking finasteride for hair loss should use contraception to prevent pregnancy and minimize this theoretical risk.
There is no proven risk associated with unprotected sex while taking finasteride. Research has shown that finasteride levels in semen are insignificant and negligible—about 650 times lower than the oral dose—and do not affect DHT levels. 12,13
Is There an Association of Finasteride and Prostate Cancer?
The prostate, a small gland found only in men, is located below the bladder and in front of the rectum, surrounding the urethra (the tube that carries urine out of the body). As men age, the prostate may enlarge, putting pressure on the urethra and causing urinary symptoms. This condition, known as Benign Prostatic Hyperplasia (BPH), is common among older men and is not cancerous.
Symptoms of BPH include:
- Frequent urination, especially at night
- Difficulty starting urination
- Weak or interrupted urine stream
- Inability to completely empty the bladder
- Sudden urgency to urinate
DHT is primarily responsible for prostate enlargement in BPH and may also contribute to prostate cancer development. Finasteride, initially FDA-approved at a 5 mg dose for BPH (five times higher than the 1 mg dose for hair loss), works by lowering DHT levels in the prostate, reducing its size, and improving urinary symptoms. While a 5 mg dose of finasteride, typically used for treating prostate enlargement, reduces prostate size by approximately 27%, the lower 1 mg dose, commonly prescribed for hair loss, can still decrease prostate size by about 20%.
While BPH is not a risk factor for prostate cancer, finasteride remains a valuable treatment for treating BPH as it may reduce the risk of developing prostate cancer while simultaneously improving urinary symptoms.14 Although finasteride is not FDA-approved for treating prostate cancer, it can aid in detecting the disease. By reducing the size of the prostate, finasteride increases the likelihood that a biopsy will find existing cancers, improving early detection.
Despite the above facts regarding finasteride and it’s positive effects on the prostate, one of the ongoing debates regarding finasteride is its potential link to prostate cancer. Prostate cancer is one of the most common cancers in men and is the second leading cause of cancer-related death in men, after lung cancer. Although all men are at risk, this risk increases significantly with age. Other contributing factors include family history, chemical exposure, and race.
Key statistics about prostate cancer:
- Prostate cancer is the second leading cause of cancer-related death in men
- A man is diagnosed with prostate cancer every two minutes
- 1 in 8 men will be diagnosed with prostate cancer during their lifetime
- In 2024, an estimated 35,250 men are expected to die from prostate cancer
- A man dies from prostate cancer every 15 minutes
The Prostate Cancer Prevention Trial (PCPT) was conducted between 1993 and 2013 with nearly 19,000 men aged 55 and older. Its purpose was to assess whether 5 mg oral finasteride (Proscar®) could reduce the risk of prostate cancer.
The 2013 results indicated that finasteride lowered the overall risk of developing prostate cancer by 30%. Though men taking finasteride had an 18% reduction in total prostate cancer cases, there was a slight increase (1.3% higher than the placebo group) in the incidence of high-grade prostate cancers. This raised concerns that finasteride might increase the risk of developing more aggressive forms of prostate cancer—not the overall risk of getting prostate cancer—prompting the FDA to issue a black box warning on the drug’s label.
However, further analysis suggested that this increase in high-grade cancers may have been due to improved detection rather than the drug itself. Finasteride enhances the sensitivity (better at spotting when something’s wrong) of the Prostate-Specific Antigen (PSA) test, which is a blood test used to help screen for prostate abnormalities, including cancer. This means it makes it more effective in detecting both overall and high-grade prostate cancers. If finasteride fails to lower PSA levels by 50%, it could be an indicator of prostate cancer. This enhanced detection may have led to an overestimation of high-grade cancer risk and an underestimation of finasteride’s benefits in reducing overall prostate cancer risk.15
Long-term studies show that finasteride does not increase the risk of death from prostate cancer. For men with low-grade cancers, the 10-year survival rate was 83% for those taking finasteride compared to 80.9% for those on a placebo. For high-grade cancers, survival rates were almost the same: 73% for finasteride users and 73.6% for placebo users. After 20 years, there was no increase in prostate cancer-related deaths among men who took finasteride. The slight increase in high-grade cancers (3.5% in the finasteride group vs. 3% in the placebo group) was not statistically significant, suggesting this difference could be due to chance.16
Is There an Association of Finasteride with Breast Cancer?
Male breast cancer is extremely rare, accounting for less than 1% of all breast cancer cases, with a prevalence of about 1 in 726 men. The average age of diagnosis ranges from 65 to 70 years old. Risk factors for male breast cancer include advanced age, genetic mutations (BRCA2), family history, hormonal imbalances, and lifestyle factors like obesity and chronic alcohol consumption.
Men with a strong family history of breast cancer or known BRCA mutations should consider routine self-exams to check for any lumps, pain, or nipple discharge.
Currently, there is no evidence that finasteride or dutasteride causes breast cancer in men. A study of 339 men with breast cancer and 6,780 men without breast cancer found no connection between the use of these 5-alpha reductase inhibitors and the development of breast cancer.
Additionally, the Prostate Cancer Prevention Trial, which evaluated nearly 19,000 men aged 55 and older who took 5 mg oral finasteride versus a placebo, found no increased incidence of breast cancer between the two groups.
To learn more about male breast cancer please click here: Cancer.org
Can Women Who Take Finasteride Get Breast Cancer?
There is currently no strong evidence that using finasteride or dutasteride increases the risk of breast cancer in women, even among those with known risk factors for the disease.
A large population-based study explored whether using these medications increases the risk of breast or gynecologic cancers in 13,853 women with female pattern hair loss. The study found no significant link between the use of these medications and an increased risk of these cancers compared to non-users. The results were consistent, with additional analyses confirming that 5α-reductase inhibitors are not associated with a higher cancer risk.
Blood Donation While Taking Finasteride
Because finasteride and dutasteride can remain detectable in the bloodstream for months after stopping treatment, users are advised to avoid donating blood for a certain period after discontinuation. The recommended deferral period for finasteride is at least one month after the last dose, while it is six months for dutasteride.
This precaution is in place to prevent the risk of a pregnant woman receiving the medication via a blood transfusion, which could theoretically affect the development of a male fetus’s genitalia.
If you are considering donating blood, it’s always a good idea to inform the blood donation center about your recent use of finasteride or dutasteride so they can provide guidance based on their protocols.
Potential Side Effects of Finasteride
Since its FDA approval in 1997, finasteride has successfully helped millions of people treat hair loss. However, concerns about potential side effects—particularly sexual side effects—along with misinformation and fears about long-term consequences can make some hesitant to use the medication. The internet is filled with personal stories about negative experiences with finasteride. While these accounts may not reflect the typical user experience, they often create fear and anxiety for those considering the medication.
Misinformation from unqualified sources, whether online or within the community, can further influence public perception and promote unjustified concerns about finasteride. Keep in mind that people are more likely to share a negative experience than a positive one when everything goes smoothly.
I aim to provide a balanced and thorough understanding of the potential side effects associated with finasteride. To do this, I will first offer insight into the prevalence of side effects unrelated to finasteride and then present clinical data from multiple studies evaluating the actual risks of the medication. This approach will help you determine whether finasteride is the right treatment option for you.
Anyone thinking about taking finasteride should talk with a hair loss specialist who knows the medication well and can answer any questions or concerns. This ensures you receive up-to-date, accurate information and guidance, helping you make a confident and well-informed decision.
Sexual Dysfunction
Sexual dysfunction in men is a common health issue that can impact various aspects of sexual performance, including reduced libido (sexual desire), erectile dysfunction, and ejaculatory disorders. The prevalence of sexual dysfunction varies based on factors such as age, underlying health conditions, medications, and lifestyle. Below is an overview of the most common types of sexual dysfunction and their typical prevalence among men.
The link between sexual function, aging, and male health was first highlighted in the early 1990s. One of the most widely cited studies, the Massachusetts Male Aging Study (MMAS), found that male sexual dysfunction—such as erectile dysfunction (ED), reduced libido, or abnormal ejaculation—commonly begins in men during their early 40s and increases with age.
The incidence of sexual dysfunction rises with each decade of life:
- 8% by age 20
- 11% by age 30
- 40% by age 40
- 50% by age 50
- 60% by age 60
- 70% by age 70
According to the National Health and Social Life Survey, 31% of younger men aged 18 to 59 reported experiencing sexual dysfunction. Additionally, data from the Massachusetts Male Aging Study (MMAS), shows that the overall prevalence of sexual dysfunction in men between the ages of 40 and 70 is 52%, with the likelihood increasing as men age.
Erectile Dysfunction (ED)
Erectile dysfunction (ED) ranges from occasional difficulty maintaining an erection to a complete inability to achieve one sufficient for sexual intercourse. It is the most common sexual disorder, affecting an estimated 30 to 50 million men in the U.S. and at least 150 million worldwide. By 2025, this number is expected to reach 322 million globally. However, these figures are likely underreported due to factors like reporting bias, cultural stigma, and a lack of inquiry into male sexual health by many physicians.
For many men, ED is a natural part of aging. However, patients taking medications like finasteride or dutasteride often blame their ED as a medication side effect. Sometimes, age alone may be the primary factor rather than the medication itself.
Factors Influencing Sexual Dysfunction in Men
Although aging is a key contributor, ED is typically multifactorial, often linked to underlying medical conditions and lifestyle factors, some of which may go undiagnosed. Healthy sexual function depends on the complex interplay of vascular, neurologic, hormonal, and psychological systems. Studies have shown that organic causes account for 15-72% of ED cases in men under 40 and increase sharply with age.17
Key factors include:
- High blood pressure: Approximately 40% of men with erectile dysfunction also have hypertension, and 35% of men with high blood pressure experience ED. 18,19,20
- Cardiovascular disease (stroke, coronary artery disease): ED can also be an early warning sign of conditions like underlying heart disease, high blood pressure, high cholesterol, low testosterone, or diabetes. Since the blood vessels in the penis are smaller than in other areas of the body, ED symptoms often manifest before more severe issues like a heart attack or stroke occur.21
A meta-analysis of 14 studies, including over 90,000 men with ED, found that these patients had a 44% higher rate of cardiovascular events, 62% more heart attacks, 39% more strokes, and a 25% increased risk of death compared to men without ED. Nearly 50% of men with diagnosed coronary artery disease also have significant ED, which often presents 3-4 years before other heart disease symptoms emerge.
- Hyperlipidemia is a medical condition characterized by elevated fat (lipids) levels in the blood, such as cholesterol and triglycerides. It is a common disorder that significantly increases the risk of cardiovascular diseases, including heart attacks, strokes, and atherosclerosis (hardening of the arteries). Genetic factors, poor diet, lack of physical activity, and other health conditions can cause hyperlipidemia. Notably, hyperlipidemia is found in approximately 42% of men with erectile dysfunction (ED), highlighting its impact on overall cardiovascular and sexual health.
- Prediabetes and Diabetes: Erectile dysfunction is strongly linked to undiagnosed prediabetes, as well as type 1 and type 2 diabetes, especially in younger adult males (ages 18 to 40). Men with ED are up to three times more likely to have undiagnosed diabetes (28%) compared to non-diabetic men with normal erectile function (10%).22,23,24 Among men over 50, those with diabetes are about twice as likely to experience ED (46%) compared to those without diabetes (24%).25 The longer a person has diabetes and the more advanced the disease, the higher their risk of developing ED.
- Obesity: Obesity is linked to a 50% higher likelihood of developing ED compared to men of average weight.26 Additionally, one-third of obese men with ED who participated in a weight loss program resolved their ED symptoms within two years.27
- Obstructive sleep apnea (OSA), defined as repetitive episodes of upper airway collapse during sleep, is a common chronic condition affecting 10% of middle-aged men.28
OSA causes repeated breathing interruptions during sleep, leading to reduced oxygen levels, impaired vascular health, and restricted blood flow—all of which directly impact the ability to achieve and maintain an erection. OSA has been linked to various conditions, including cardiovascular disease, stroke, and hypertension. One study found that 69% of men with OSA had some degree of ED.
- Hormonal Imbalances: Low Testosterone and Thyroid Conditions: One-third of diabetic men are likely to have low testosterone, which may help explain the strong link between diabetes and ED.29 Additionally, up to 35% of all men with ED are found to have low testosterone, and about 6% present with abnormal thyroid function.30,31 While testosterone deficiency can negatively affect erectile function, vascular disease and diabetes are far more common causes of ED.32
A study by Marberger et al. examined this relationship and found that sexual dysfunction was more pronounced in men with total testosterone levels below 224 ng/dL. Moreover, men with lower baseline testosterone experienced more adverse sexual effects compared to those with normal testosterone levels. This suggests that measuring baseline testosterone levels can be helpful when considering treatment with DHT-blockers in patients suspected of having low testosterone. If treatment is necessary, proper counseling should be provided before prescribing. - Urological Conditions — Benign Prostatic Hyperplasia (BPH) with Lower Urinary Tract Symptoms (LUTS): There is a strong correlation between BPH with LUTS and erectile dysfunction (ED). Up to 72% of men with symptomatic BPH will also experience ED.33,34,35
- Neurological Conditions:
- Multiple Sclerosis
- Traumatic injuries (such as spinal cord injuries)
- Medications: One reason ED becomes more common with age is that older men are more likely to be on medications, many of which list ED as a side effect. With approximately 60% of the population using prescription drugs each year, medication-induced ED has become a significant and common cause of the condition.36 Notably, an estimated 25% of all ED cases are related to medication use and may be reversible.37
Of the 12 most commonly prescribed medications in the US, eight list erectile dysfunction as a possible side effect.
These medications include:
- Blood pressure medications: amlodipine, clonidine, propranolol, metoprolol, and certain diuretics (hydrochlorothiazide, chlorthalidone, and indapamide)
- Heart failure medications: Digoxin (Lanoxin) and Spironolactone (Aldactone)
- Cholesterol-lowering medications: simvastatin and atorvastatin
- Mental health disorder medications: Antidepressants, antipsychotics, anti-anxiety, and panic disorder medications
- Dermatologic medications: Isotretinoin (Accutane)
- Antihistamines
- GERD (gastroesophageal reflux disease) medications
- Anti-seizure medications
- Opioids: Oxycodone
- Parkinson’s disease medications
- Hair loss medications
- Lifestyle Factors: While medical conditions are well-known contributors to ED, lifestyle habits, often overlooked, also play a significant role.
These include:
- Diet and Nutrition: A diet high in saturated fats, processed foods, and sugars can lead to conditions such as obesity, hypertension, and high cholesterol, all of which are risk factors for ED. Whereas a balanced diet rich in fruits, vegetables, whole grains, and healthy fats (such as those found in the Mediterranean diet) supports cardiovascular health and reduces the risk of ED.
- Physical Activity: Regular exercise improves blood circulation, lowers blood pressure, and helps maintain a healthy weight—all essential for preventing ED. Physical activity also increases nitric oxide production, which is critical for achieving and maintaining erections by promoting blood flow to the penis.
- Smoking and Excessive Alcohol Use: Smoking damages blood vessels and impairs circulation, making it a major risk factor for ED. Research shows that smokers who quit show a 25% improvement in erectile function after one year.38 Alcohol, especially when consumed in excess, can lead to penile nerve damage, hormonal imbalances, and reduced blood flow, all of which contribute to ED. More specifically, heavy drinkers are more likely to experience erectile problems than non-drinkers or moderate drinkers. 39
- Poor Sleep Quality: Sleep is essential for maintaining hormonal balance, including the production of testosterone, which is necessary for healthy sexual function. Testosterone is predominantly produced during sleep, especially during REM (rapid eye movement) cycles. Poor sleep quality or sleep disorders like sleep apnea can reduce testosterone levels, which may lower libido and contribute to erectile dysfunction (ED).
Irregular sleep patterns or shift work can disrupt circadian rhythms, affecting hormone release and nitric oxide production—both necessary for sexual performance. Chronic sleep deprivation can also increase stress and cortisol levels, further hindering sexual function. A study found that men who slept less than 5 hours per night had reduced testosterone levels (10% to 15%). This condition is experienced by at least 15% of the US working population. By comparison, normal aging is associated with a decrease of testosterone levels by 1% to 2% per year, after the age of 30. Chronic sleep deprivation is linked to fatigue, poor mood, and diminished sexual interest, contributing to ED. - Excessive Use of Pornography: Some researchers propose that the abuse of pornography may contribute to erectile dysfunction (ED). The theory suggests that repeated exposure to highly stimulating and varied sexual content could desensitize the brain’s reward system, making it more challenging for some men to become aroused in real-life sexual situations. In one study of 3,419 men aged 18 to 35, 21.48% experienced some degree of ED
- Mental Health Disorders: Anxiety, depression, and stress can significantly reduce libido and contribute to sexual performance issues. Men with erectile dysfunction (ED) are 2.5 times more likely to experience anxiety and 3.5 times more likely to suffer from depression.40 Additionally, men with depression are almost 40% more likely to develop ED compared to those without depression, while the prevalence of depression in men with ED is nearly three times higher than in the general population 41
Even when the root cause is physical, ED often leads to psychological consequences, including marital and relationship problems, cultural expectations, loss of self-esteem, shame, anxiety, and depression. These emotional effects can significantly impact both the patient and their partner, reducing overall quality of life.
Fortunately, ED is almost always treatable. Identifying whether the cause is psychological or organic is essential for determining the appropriate treatment. Improvements in diet, exercise, sleep, alcohol and stress reduction (through therapy, meditation, and relaxation techniques) have been clinically proven to enhance sexual function.
Low Libido (Decreased Sexual Desire)
Studies show that 15-25% of men may experience low libido at some point in their lives. It is more common as men age but can affect younger men as well due to factors such as stress, mental health issues, chronic diseases, hormonal imbalances (like low testosterone), or medications (the same ones that may cause ED).
Ejaculatory Disorders
The effect on sperm production was found to be below the level considered critical by researchers, meaning that these changes are unlikely to impact male fertility significantly.42 However, individual responses to these medications can vary from person to person.
Decreased Semen Volume (Oligospermia)
Decreased semen volume can cause semen to become thin and watery. This occurs when a man has a low sperm count (oligospermia), which is typically fewer than 15 million sperm cells per milliliter of semen.
This can occur with the use of finasteride because the medication reduces the size of the prostate, a small gland responsible for producing a portion of the seminal fluid. While this effect is beneficial for treating benign prostatic hyperplasia (BPH) in men with normal-sized prostates, it can lead to a reduction in the amount of semen produced. Studies have shown that this decrease in semen volume is a possible side effect of finasteride.43
For more information, please see Infertility Concerns with Finasteride.
Delayed Ejaculation
Delayed ejaculation is an uncommon condition affecting about 1-4% of men, where it becomes challenging to reach ejaculation during sexual activity despite normal arousal and stimulation. It is more frequently seen in older men. While it is less common than other sexual issues, such as erectile dysfunction or premature ejaculation, it can still lead to significant emotional stress and strain on relationships.
A variety of factors can cause delayed ejaculation. Psychological conditions such as anxiety or stress are common contributors. Medications, particularly antidepressants like selective serotonin reuptake inhibitors (SSRIs), can also play a role. Nerve damage from conditions such as diabetes may lead to delayed ejaculation, as can excessive alcohol or drug use. Hormonal imbalances, like low testosterone levels, can also contribute to the condition. Additionally, prostate issues, including surgery or treatments related to the prostate, may affect ejaculation.
Studies on Sexual Side Effects of Finasteride
Sexual side effects are possible for men taking finasteride (1 mg for hair loss or 5 mg for an enlarged prostate) and dutasteride (0.5 mg). These side effects may include reduced sex drive (libido), difficulty getting or maintaining an erection (erectile dysfunction), and ejaculation issues. In most cases studied during clinical trials, these sexual side effects appeared within the first year of treatment. However, they often got better over time, even for men who continued taking the medication.
In a study of men with AGA who took 1mg of oral finasteride daily, the overall effect of hair growth was seen in 87.1%, with 23 out of 3,177 patients having sexual side effects (0.7%).44
In clinical trials using placebo pills (which do not impact DHT levels), around 0.7% of men reported experiencing side effects like erectile dysfunction and decreased libido. This indicates that about 0.7% of men may experience these types of sexual dysfunction naturally, even without taking medication. As men get older, particularly after age 70, the likelihood of sexual dysfunction increases significantly, with estimates suggesting that between 50% to 100% of men in this age group may experience some form of sexual dysfunction.45,46
In this 2006 study, 2% of 708 men with AGA who received finasteride 1mg daily for four months developed a sexual side effect.
Research shows that sexual side effects occur in 2.1% to 3.8% of people, with erectile dysfunction being the most common. This is followed by ejaculatory issues and a decrease in sexual desire (libido). These effects typically happen early in the treatment and often improve either when the medication is stopped or with continued use over time.
Notably, a review of clinical trials studying finasteride for hair loss (androgenic alopecia) found that the reports of side effects were only sometimes reliable. The data was often of poor quality, biased, and didn’t fully reflect what patients might experience in everyday use. Additionally, most people in these studies had only been on finasteride for a year or less, making it hard to understand the long-term effects.
In the first year of taking 1 mg finasteride, some men reported sexual side effects, though they were relatively rare. These included:
- Decreased sex drive (reported by 1.8% of men on finasteride compared to 1.3% on placebo)
- Erectile dysfunction (1.3% on finasteride vs. 0.7% on placebo)
- Ejaculation disorders (1.2% on finasteride vs. 0.7% on placebo)
- Reduced semen volume (0.8% on finasteride vs. 0.4% on placebo)
- Only 1.2% stopped taking finasteride due to these side effects, and they generally resolved once the medication was discontinued.
No other significant side effects were observed in the studies. Notably, many men who continued using finasteride saw improvements, with 58% reporting that the side effects disappeared over time. After stopping the medication, side effects typically disappear within a few weeks.
In the Finasteride Male Pattern Hair Loss Study, researchers conducted two 1-year studies involving 1,553 men aged 18 to 41 with male pattern hair loss. These studies were double-blind, meaning neither the participants nor the researchers knew who received the medication or placebo. During the first year, 4.2% of men taking finasteride reported sexual side effects, compared to 2.2% in the placebo group. Only a few men stopped the study due to these side effects—11 men (1.4%) in the finasteride group and eight men (1.0%) in the placebo group. Importantly, the side effects went away after stopping finasteride.47
A long-term study found that sexual side effects such as decreased libido, erectile dysfunction (ED), and ejaculatory disorders occurred in fewer than 2% of the 1,215 men involved. These side effects went away not only in men who stopped taking the medication but also in most of those who continued treatment. By the fifth year of taking finasteride, the incidence of these side effects dropped to 0.3% or less. The occurrence of side effects was similar to that seen in the placebo group, both after one year and five years of treatment.
A 48-week study involving 212 men aged 18 to 40 with androgenetic alopecia (AGA) looked at the effects of taking 1 mg of finasteride daily. The study found that 1.9% of men in the finasteride group experienced sexual side effects, compared to 0.9% in the placebo group. Importantly, no one dropped out of the study due to these side effects. Of the two men who did report sexual issues, one found that the symptoms improved while still on the treatment, and the other saw his symptoms disappear two weeks after stopping the medication.
Another study by Roberts et al. looked at different doses of finasteride (5 mg, 1 mg, 0.2 mg, and 0.01 mg) to treat hair loss (androgenetic alopecia) in men aged 18 to 36. After six months, researchers checked for sexual side effects like decreased libido and erectile dysfunction. The results showed no significant differences in the occurrence of these side effects between the different doses and the placebo group. For example, the rates were similar: 3.6% for the 5 mg dose, 4.3% for the 1 mg dose, and 3.4% for the placebo. All of the differences in these numbers were considered insignificant.
In a study by Drake et al., researchers compared different daily doses of finasteride (0.01 mg, 0.05 mg, 0.2 mg, 1 mg, and 5 mg) to a placebo in 249 men with androgenetic alopecia. The study found that the only side effects reported by more than one person were headaches and decreased libido (sex drive). Interestingly, the placebo group had a 4.5% rate of reduced libido, while the group taking 1 mg of finasteride reported 0%, and the 5 mg group had a 2.6% rate.
A case-control study found that men taking 1 mg of finasteride daily had no differences in sexual or erectile function compared to those who were not taking the medication. Additionally, an observational study showed that sexual function in men did not decrease during the first 4 to 6 months of finasteride use.
In a 48-week randomized, double-blind study, researchers compared the effectiveness of 1 mg and 0.2 mg of finasteride in 414 men with hair loss (androgenetic alopecia). The study found that decreased libido occurred in 2.9% of men taking 1 mg, 1.5% of men taking 0.2 mg, and 2.2% of those in the placebo group. Most cases of decreased libido improved while continuing the treatment, and no one stopped the study due to these side effects.
In a one-year study by Leyden et al., 326 men with male pattern hair loss were randomly assigned to take either 1 mg of finasteride daily or a placebo. After the initial year, the study continued for another year. About 2% of men in both the finasteride and placebo groups reported sexual side effects like reduced libido, with two men in each group mentioning this symptom. Additionally, one man in the finasteride group experienced impotence. Importantly, no one stopped the study due to sexual side effects, and there were no increases in drug-related issues during the extended follow-up period.
Depression
In the U.S., nearly 21% of adults, or 50 million Americans, live with mental illness, and about 8.5% (or 21 million adults) have experienced a major depressive episode. Adult women are more likely to experience major depressive episodes compared to men, with a prevalence of 10.5% among women versus 6.2% among men, as reported by the National Institute of Mental Health.
Depression is particularly common among young people, with over 10% experiencing symptoms severe enough to disrupt their daily activities, including school or work. Young adults aged 18 to 25 are at the highest risk of developing depression. The COVID-19 pandemic worsened mental health conditions, with the percentage of adults diagnosed with anxiety or depression increasing from 36.4% to 41.5% between August 2020 and February 2021. The most significant rise was seen in adults aged 18 to 29.48
Depression can be caused by a mix of genetic, biological, environmental, and psychological factors. Genetics, such as a family history of depression, can increase the risk, while biological factors include changes in brain chemistry or hormones. Environmental factors like stressful life events, trauma, substance abuse, or ongoing challenges such as financial or chronic health problems can also play a role. Psychological factors, including personality traits, negative thinking patterns, or low self-esteem, can make some people more vulnerable to developing depression.
Certain medications may list depression as a potential side effect.
They include:
- Acid reflux medications like omeprazole, esomeprazole, ranitidine, and famotidine
- Allergy medications like montelukast and cetirizine
- Blood pressure medications like atenolol, metoprolol, enalapril, and quinapril
- Cholesterol-lowering medications
- Anxiety medications like alprazolam, diazepam, and lorazepam (and the sleep medication zolpidem)
- Pain medications like ibuprofen, cyclobenzaprine, hydrocodone, and tramadol
- Birth control and hormone therapy, which includes anything containing estrogen
- Antiseizure medications like gabapentin, topiramate, and lamotrigine
- Isotretinoin (Accutane)
- Acyclovir
This study revealed that 37% of participants used at least one prescription medication that listed depression as a potential side effect. For people not taking any medications with depression as a side effect, the likelihood of having depression was 5%, regardless of how many other medications they were on. However, the risk of depression rose significantly with each additional medication that listed depression as a side effect. For people taking two medications with depression as a side effect, they were twice as likely to develop depression, while those on three had three times the risk.
Even among people already taking antidepressants, a similar pattern was noticed. For those taking an antidepressant and no other medications with depression as a potential side effect, the risk of experiencing depression was 14%. This risk increased to 18% for those on one additional medication, 27.5% for two additional medications, and 28% for those taking three or more medications.49
In some cases, medications like finasteride and dutasteride have been linked to reports of depression and anxiety, though these side effects are rare, occurring in about 1-2% of users. While the connection between finasteride/dutasteride and depression, particularly in its oral form, is still under study, some research suggests that reducing dihydrotestosterone (DHT) levels with finasteride might disrupt hormone balance and neurosteroid activity, both of which are important for mood regulation. However, Basaria et al. found no signs of androgen (testosterone, DHEA-S) deficiency, reduced androgen activity, or continued inhibition of the enzyme 5-alpha reductase in men who reported persistent sexual symptoms after using finasteride. However, these individuals did exhibit symptoms of depressed mood, and brain imaging (fMRI) results were similar to patterns commonly seen in individuals with depression. This suggests that while hormonal imbalances may not be the cause, mood-related changes in brain function could play a role in these ongoing symptoms.
Can Finasteride Lead to Depression?
A more recent meta-analysis found that younger men taking 1 mg of oral finasteride may be more prone to psychological side effects, particularly because they are often more concerned about issues like sexual dysfunction, which can already heighten anxiety related to hair loss. This anxiety may make them more vulnerable to mental health challenges such as depression and anxiety.
Understanding the Nocebo Effect
The nocebo effect happens when negative beliefs or worries about a medication lead someone to feel or report side effects, even if the medication isn’t causing them.
The nocebo effect can play a significant role in medications like finasteride and dutasteride. If patients are overly concerned or have read about possible side effects—especially from online forums or anecdotal stories—they may be more likely to notice these symptoms, even if the medication isn’t causing them. Statistically, they are more likely to experience these effects simply due to their expectations. This is the nocebo effect at work.
Understanding the nocebo effect is important because it shows how a patient’s mindset can influence their experience with a medication. This is especially relevant for drugs like finasteride and dutasteride, where fear of side effects may cause some patients to stop treatment early or avoid the medication altogether, possibly missing out on its benefits.
Healthcare providers can help reduce the nocebo effect by:
- Explaining the actual risks and benefits of the medication
- Talking openly with patients about any concerns or fears related to potential side effects
- Encouraging patients to focus on the expected benefits rather than the risks
Addressing these concerns can help patients make more informed choices about their treatment and increase their chances of experiencing the full benefits of medications like finasteride.
Nocebo Effect Studies
In this study, the group that was informed about the potential sexual side effects of finasteride reported a higher rate of erectile dysfunction (ED) compared to the group that wasn’t given this information. The side effects disappeared completely within five days after stopping the medication, which shows that the nocebo effect can play a role in causing side effects. This also highlights the influence of psychological factors on how patients experience symptoms.
A 2017 study followed 4,284 men who took 1 mg of finasteride daily for four years. Among those who had never experienced sexual dysfunction before, only 34 men (0.79%) reported new sexual dysfunction that continued after stopping finasteride. Additionally, of the 103 younger men who experienced sexual dysfunction, 34 reported that the issue became persistent. Researchers suggested that the nocebo effect likely played a significant role in developing these side effects. This study also highlights that decreased sex drive, like other side effects, may be influenced by the nocebo effect. However, the primary focus of the study was on erectile dysfunction, as it is easier to measure and track compared to other sexual side effects. 50,51
Kaufman et al. found that adverse reactions related to sexual functions most commonly occurred in year 1 of treatment with finasteride and placebo (4.4% vs. 2.2%); on the contrary, the incidence of adverse reactions decreased in year 5 (0.6% vs. 0% for finasteride and placebo, respectively).
Topical Finasteride Side Effects
Topical finasteride is believed to reduce the risk of systemic side effects by primarily targeting 5-alpha-reductase activity directly in the scalp. This localized action limits the amount of the drug that enters the bloodstream, thus lowering its overall concentration and minimizing potential side effects that are more common with oral administration.52,53 Additionally, the concentration of finasteride in the blood is 100 times lower with topical application. After 24 weeks of use, bloodstream DHT levels were reduced by 34.6% with topical finasteride compared to a 55.6% reduction with oral finasteride, demonstrating that while both forms lower DHT, topical finasteride has a much milder systemic effect.
Mild side effects reported with topical finasteride include skin irritation, itching, or redness. In rare cases, some systemic side effects, such as decreased libido and erectile dysfunction, may still occur.
Persistent Side Effects + Post-Finasteride Syndrome (PFS)
Research indicates finasteride’s side effects are generally mild, rare, and reversible. These side effects are not permanent and usually go away over time. Most patients find that it takes a couple of weeks, while others needed a few months for any lingering side effects to resolve, although this can vary depending on how your body responds to the medication and any stress you may have. The time it takes for side effects to fully disappear can feel prolonged, depending on your body’s recovery process. If you notice side effects, don’t try to guess or rely on Google—just check in with your doctor. They’re the best person to guide you.
Post-Finasteride Syndrome (PFS)
Post-finasteride syndrome encompasses a range of adverse effects that continue despite the discontinuation of finasteride or dutasteride. These adverse effects are characterized by persistent sexual, neurological, and psychological symptoms.
The persistence of sexual side effects from finasteride is rare. So far, no studies have conclusively proven a direct connection between finasteride and these persistent side effects, and the overall frequency of these events hasn’t been established.54,55 It remains unclear whether these cases are directly caused by the medication or influenced by other factors, such as the naturally high rates of sexual dysfunction in the general population, pre-existing medical conditions, or the nocebo effect (where negative expectations can cause perceived side effects). Because many of these patients haven’t undergone thorough medical and psychological evaluations, it’s challenging to determine other potential causes.
Furthermore, (PFS) is controversial and not universally recognized by all medical professionals, yet it contributes to concerns surrounding the drug. Ongoing research aims to understand PFS better, though some criticize the existing studies for their poor quality.
The current scientific literature is divided on the issue. Studies that reported a higher occurrence of persistent side effects have been criticized for biases, small sample sizes, and inconclusive results. They also fail to distinguish whether the symptoms are the result of actual drug side effects or a psychological response (nocebo effect). One study found that 43.6% of patients who were warned about potential sexual side effects before taking finasteride reported symptoms, compared to only 15.3% of those who were not informed, highlighting the role of the nocebo effect.56
The prevalence of persistent side effects remains unknown, and the scientific community has not yet fully recognized PFS.57,58 While PFS may exist, it seems to affect only certain individuals who may be more sensitive to the drug, even when exposed to low doses for a short time, and can persist for a long period.59
Despite these concerns, earlier studies have demonstrated the safety of finasteride. For example, a large trial in 2003 found that sexual side effects were more persistent in the placebo group than in those treated with finasteride, suggesting that the drug may not have been responsible for the complaints in these cases.
A systematic review and meta-analysis from 2016 found that patients taking finasteride with benign prostatic hyperplasia (BPH) had a significantly higher risk of sexual dysfunction compared to those being treated for androgenetic alopecia (AGA). The difference in these findings was likely due to two key factors: the average age of the patients and the daily dose of the medication used for each condition.
A 2019 study found that individuals with a history of sexual or psychological conditions are at a higher risk of experiencing adverse reactions to finasteride, and these reactions may be more severe. Therefore, it is critical to assess for pre-existing sexual dysfunction and mental health issues before starting finasteride to manage potential side effects better.
The side effects of topical finasteride are usually mild and can include scalp irritation, itching, or redness. Research indicates that topical finasteride typically causes fewer systemic side effects than the oral version, as it is absorbed primarily through the scalp, reducing its impact on the body’s overall DHT levels. However, some studies have still reported cases of sexual side effects, such as decreased libido and erectile dysfunction, with topical use. While these side effects are less frequent with topical finasteride compared to the oral form, they cannot be entirely avoided because the topical solution still lowers blood stream DHT levels to some extent.
If you have a history of depression, sexual dysfunction, or infertility, it’s important that you discuss your conditions with your hair loss specialist. They can help assess the risks and benefits specific to your situation and explore alternative treatment options if sexual side effects become a concern.
Finasteride Side Effects for Women
While rare, some of the side effects include rash, headache, dizziness, menstrual irregularities, and decreased libido.
Since topical finasteride is applied directly to the scalp, much less of the drug is absorbed into the bloodstream than oral. This localized application helps limit systemic exposure, reducing the likelihood of widespread side effects.
Who Should Not Use Finasteride:
- Anyone under the age of 18 is not approved to use finasteride
- Women who are pregnant or may become pregnant should avoid using finasteride. Women of childbearing age who are sexually active and are prescribed finasteride should use effective contraception
- Men with severe liver disease may not metabolize finasteride properly because the drug is processed in the liver
Your Experience Is Your Own
Medications don’t affect everyone the same way—and that’s completely normal.
Some people may experience side effects, while others may not. Even if side effects do happen, how strong they are—and how long they last—can be different for each person.
Be Careful with Online Stories
Just because someone online had a tough time doesn’t mean you will too. Think of it like online reviews: people are more likely to post about a bad experience than a good one. That doesn’t mean the treatment is bad—it just means the loudest voices aren’t always the most common ones.
If you have questions or concerns, talk to your hair loss specialists instead of relying on forums or social media. Your care should be based on facts, not fear.